Easy Inquiries with Wi.Tec
Gas measurements are a complex matter. For your inquiry, please use our super simple application questionnaire or send us an informal email to get in touch with us quickly.

Gas measurements are a complex matter. For your inquiry, please use our super simple application questionnaire or send us an informal email to get in touch with us quickly.
Wi.Tec-Sensorik · The Gas Measurement Company

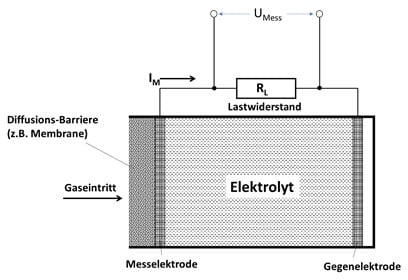
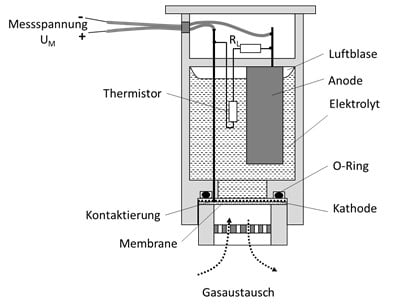
Der O2.sens von Wi.Tec basiert auf einem elektrochemischen Verfahren. Bei diesem Verfahren findet eine chemische Reaktion mit dem zu messenden Sauerstoff und einem flüssigen Elektrolyten statt. Bei dieser Reaktion werden pro Sauerstoffmolekül 4 Elektronen freigesetzt. Der daraus resultierende Sensorstrom IM oder auch die Messpannung UM steigt proportional mit der Anzahl der reagierenden O2-Moleküle an. Somit erhält man eine Sensorkennlinie, die mit steigender O2-Konzentration linear ansteigt.
Der Flüssigelektrolyt befindet sich in einer abgeschlossenen Kammer, die gasseitig mit einer durchlässigen Membrane abgedichtet ist. Durch diese Membrane gelangt der Sauerstoff zum Elektrolyten und führt dann zu dem beschriebenen Messeffekt. Bei dieser Reaktion verbraucht sich das Anodenmaterial, sodass die Lebensdauer dieses Sensors limitiert ist. Eine Membrane mit einer hohen Durchlässigkeit führt zwar zu einer hohen und schnellen Signaländerung (=kurze Ansprechzeit), aber auch zu einer kürzeren Lebensdauer, da pro Zeit mehr chemische Reaktionen stattfinden. Membranen mit einer geringen Durchlässigkeit zeigen somit ein umgekehrtes Verhalten.